Researchers shrink oven-size machine for diagnosing and halting the spread of cancer to something worn on the wrist

A new wearable device, tested on animals, can capture and remove tumor cells circulating in the bloodstream. With further development, the blood-filtering gadget could be used to diagnose, and perhaps treat, metastatic cancer in humans.
Inventors of the microfluidic device, at the University of Michigan, described it last week in the journal Nature Communications.
Catching cancerous cells circulating in the bloodstream could save lives. Cancer spreads when cells from primary tumors break off and travel through the bloodstream and the lymph system. Most circulating tumor cells, or CTCs, that enter the bloodstream die, but some can travel to new areas of the body to settle and form tumors.
It is this spreading of cancer, or metastasis, that primarily causes cancer-related deaths. “These circulating tumor cells are the seeds of metastasis,” says Sunitha Nagrath, an associate professor of chemical engineering at the University of Michigan who co-authored the paper.
So Nagrath and her team have built a wearable microfluidic machine capable of identifying cancer cells and nabbing them. When blood drawn from a vein is pumped through the device, it captures tumor cells and then pumps the blood back into the body.
In addition to its envisioned purpose as a cancer treatment that filters out tumor cells, the microfluidic machine could be used in a diagnostic role. The researchers say it could estimate the number of circulating tumor cells in the bloodstream, or help characterize the tumor from which they came.
“Our goal is to establish this blood biopsy concept as a routine clinical test,” says Nagrath. “There’s a long way to go, but eventually it could be used in a setting where early-stage cancer patients who are undergoing surgery [to remove tumors] could wear this device and get as many tumor cells out of their blood as possible.”
More than a hundred devices designed to capture or monitor circulating tumor cells have been proposed, but none of them can filter large volumes of blood, Nagrath says. Instead, these devices are designed to accept single draws of about a tablespoon of blood, which provides only a snapshot of what might be circulating in the bloodstream.
“That doesn’t represent what’s happening in the entire blood,” says Nagrath. The one device approved by the U.S. Food and Drug Administration to perform this type of analysis is about the size of an oven, she says.
Nagrath’s team has shrunk that machinery to about the size of a cigarette box, and equipped it to handle more blood. In tests performed on dogs with cancer, Nagrath’s device filtered about 1 to 2 percent of the animals’ entire blood volume in about two hours. By doing so, it captured 3.5 times as many tumor cells per milliliter of blood than was possible with a series of individual blood draws, the study authors report.
Nagrath and her colleagues acknowledge that the blood volume that passed through the prototype device in two hours is still too small. But the team says they hope to increase the throughput of the device so that all of the blood in the human body can run through it in two to three hours. “We have some ideas about how we want to accomplish that,” Nagrath says.
In the current prototype, tested on dogs, blood is filtered through graphene oxide nanosheets equipped with antibodies that bind to the surface of cancer cells. The system also continually injects a drug called Heparin to prevent blood from clotting while in the device. Finally, the blood—minus the cancer cells that are stuck to the nanosheets like flies on flypaper—is pumped back into the body. The device contains an on-board power source and microcontroller that communicates wirelessly.
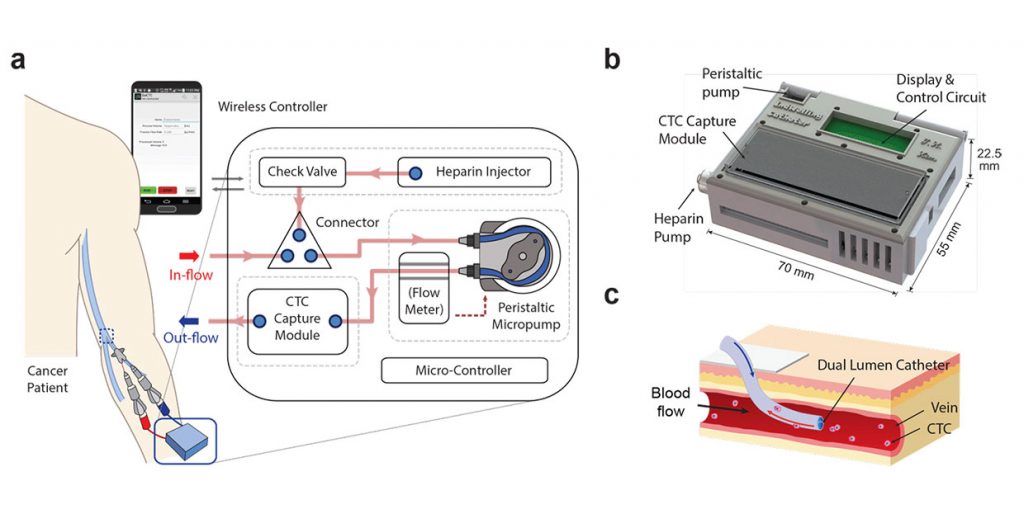
A closer look at how the wearable device would function when connected to a human
Further animal studies are required before the device can be tested on humans, Nagrath says. She and the team aim to reach the human trial stage within the next three years.
source: www.spectrum.ieee.org





















































